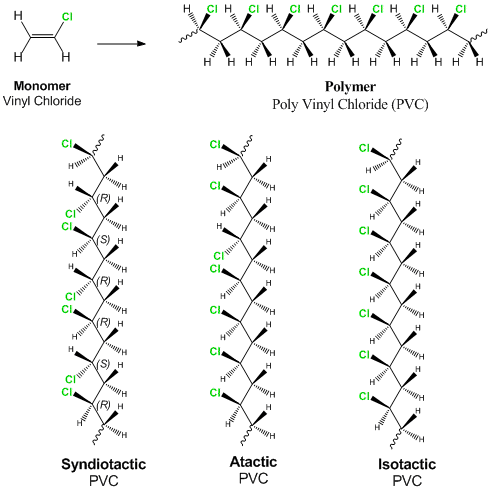
Poly(ethylene glycol) (PEG) and poly(vinyl chloride) (PVC) are examples of polymers, large organic molecules composed of repeating smaller units covalently bonded together. Polymers have very different properties depending (in part) on their
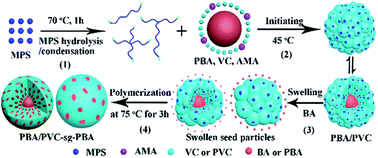
Morphology and film performance of phthalate-free plasticized poly(vinyl chloride) composite particles via the graft copolymerization of acrylate swelling flower-like latex particles - RSC Advances (RSC Publishing)

Polymerization of vinyl acetate gives poly(vinyl acetate). Hydrolysis of this polymer in aqueous sodium hydroxide gives the useful water-soluble polymer poly(vinyl alcohol). Draw the repeat units of both poly(vinyl acetate) and poly(vinyl

Riboflavin Surface Modification of Poly(vinyl chloride) for Light-Triggered Control of Bacterial Biofilm and Virus Inactivation | ACS Applied Materials & Interfaces

Statement: Vinyl chloride is a monomer that can be used for the preparation of the polyvinyl chloride. If true enter 1 else 0.

Synthesis of poly(vinyl acetate)‐graft‐polystyrene by a combination of cobalt‐mediated radical polymerization and atom transfer radical polymerization - Kaneyoshi - 2007 - Journal of Polymer Science Part A: Polymer Chemistry - Wiley Online

PDF) Poly(vinyl alcohol) Polymers with a Low Degree of Hydrolysis. II. Complex Formation with Ammonium Laurate and Sodium Lauryl Sulfate | Geir Braathen - Academia.edu

Cross-Linked Poly(vinylbenzyl chloride) Anion Exchange Membranes with Long Flexible Multihead for Fuel Cells | ACS Applied Energy Materials

Desorption of hydrolyzed poly(AM/DMDAAC) from bentonite and its decomposition in saltwater under high temperatures
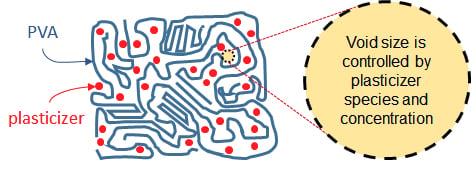
Polymers | Free Full-Text | The Impact of Plasticizer and Degree of Hydrolysis on Free Volume of Poly(vinyl alcohol) Films

Dechlorination of Poly(vinyl chloride) Wastes via Hydrothermal Carbonization with Lignin for Clean Solid Fuel Production | Industrial & Engineering Chemistry Research

EP0339371A2 - Method for preparing poly(vinyl alcohol)-co-poly(vinylamine) via a two-phase process - Google Patents
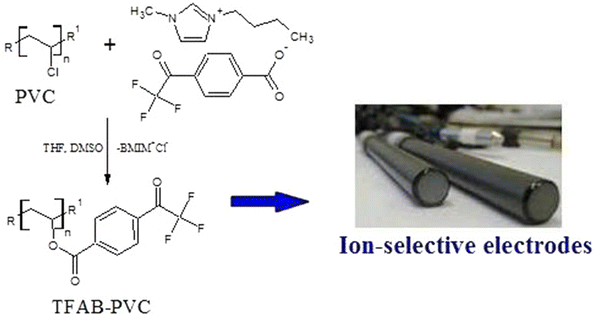
Chemically modified (poly)vinyl chloride with built-in neutral carrier function as a new material for ion selective electrodes | SpringerLink




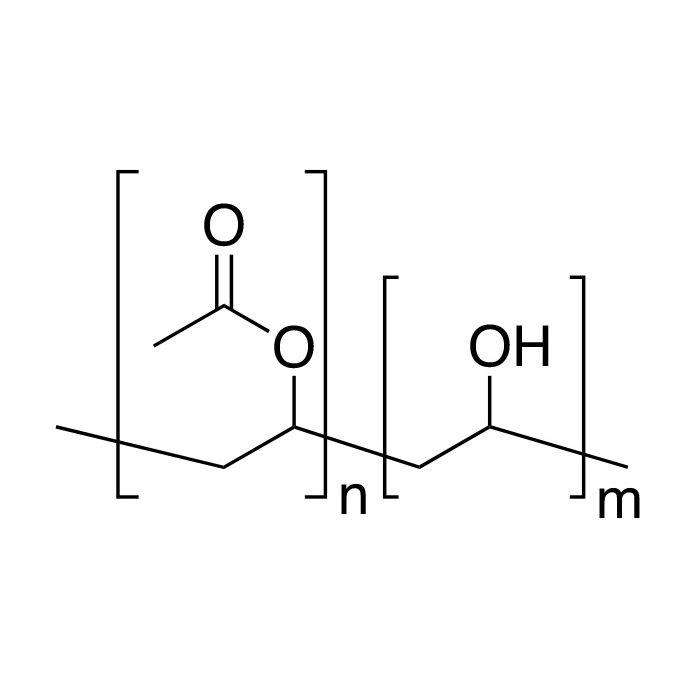


![Poly(vinyl alcohol) [88 mol.% hydrolyzed] | Polysciences, Inc. Poly(vinyl alcohol) [88 mol.% hydrolyzed] | Polysciences, Inc.](https://www.polysciences.com/media/catalog/product/cache/c44c46ddedcd391a2ef31315a65fb4d0/1/5/15132_1.jpg)